| Kidney Res Clin Pract > Volume 34(2); 2015 > Article |
|
Abstract
Background
Peritoneal fibrosis is one of the major causes of technical failure in patients on peritoneal dialysis. Epithelial-to-mesenchymal transition (EMT) of the peritoneum is an early and reversible mechanism of peritoneal fibrosis. Human peritoneal mesothelial cells (HPMCs) have their own reninŌĆōangiotensinŌĆōaldosterone system (RAAS), however, it has not been investigated whether aldosterone, an end-product of the RAAS, induces EMT in HPMCs, and which mechanisms are responsible for aldosterone-induced EMT.
Methods
EMT of HPMCs was evaluated by comparing the expression of epithelial cell marker, E-cadherin, and mesenchymal cell marker, ╬▒-smooth muscle actin after stimulation with aldosterone (1ŌĆō100nM) or spironolactone. Activation of extracellular signal-regulated kinase (ERK)1/2 and p38 mitogen-activated protein kinase (MAPK) and generation of reactive oxygen species (ROS) were assessed by western blotting and 2ŌĆ▓,7ŌĆ▓-dichlorofluororescein diacetate staining, respectively. The effects of MAPK inhibitors or antioxidants (N-acetyl cysteine, apocynin, and rotenone) on aldosterone-induced EMT were evaluated.
Results
Aldosterone induced EMT in cultured HPMCs, and spironolactone blocked aldosterone-induced EMT. Aldosterone induced activation of both ERK1/2 and p38 MAPK from 1 hour. Either PD98059, an inhibitor of ERK1/2, or SB20358, an inhibitor of p38 MAPK, attenuated aldosterone-induced EMT. Aldosterone induced ROS in HPMCs from 5 minutes, and antioxidant treatment ameliorated aldosterone-induced EMT. N-acetyl cysteine and apocynin alleviated activation of ERK and p38 MAPK.
Conclusion
Aldosterone induced EMT in HPMCs by acting through the mineralocorticoid receptor. Aldosterone-induced generation of ROS followed by activation of ERK, and p38 MAPK served as one of the mechanisms of aldosterone-induced EMT of HPMCs.
Keywords
Aldosterone, Epithelial-to-mesenchymal transition, Mesothelial cell, Peritoneal dialysis, Peritoneal fibrosisLong-term peritoneal dialysis (PD) results in peritoneal damage that is characterized by a decreased ultrafiltration capacity associated with submesothelial fibrosis, accumulation of extracellular matrix, and neoangiogenesis [1ŌĆō3].
Recent data have revealed that peritoneal mesothelial cells play an important role in peritoneal fibrosis via phenotype transition and production of extracellular matrix. Y├Ī├▒ez-M├│ et al [4] found that mesothelial cells isolated from dialysate effluents showed phenotypic transition. This phenomenon of epithelial-to-mesenchymal transition (EMT) denotes loss of epithelial characteristics and acquisition of a fibroblast-like phenotype, and is suggested as a key process in the onset and progression of peritoneal fibrosis.
There are several lines of evidence that the reninŌĆōangiotensinŌĆōaldosterone system (RAAS) is involved in organ fibrosis [5ŌĆō8]. Activation of the RAAS is related to the development of tubulointerstitial fibrosis in the kidney, and hepatic and lung fibrosis [6,9,10]. Aldosterone, a final end-product of RAAS, is reported to be an important mediator of cardiac fibrosis [11], and spironolactone ameliorates peritoneal fibrosis in animal models of peritoneal fibrosis and inflammation [12].
To determine the effect of aldosterone on peritoneal EMT, we investigated whether aldosterone induced EMT of peritoneal mesothelium and examined the mechanism of aldosterone-induced phenotypic transition of the peritoneum.
All chemicals and tissue culture plates were purchased from SigmaŌĆōAldrich (St. Louis, MO, USA) and Nunc Labware (Waltham, MA, USA), unless otherwise stated.
Human peritoneal mesothelial cells (HPMCs) were isolated from a piece of omentum, obtained from consenting patients receiving elective abdominal surgery according to the method described by Stylianou et al [13]. HPMCs were identified by phase-contrast microscopy according to morphological criteria and by immunofluorescence staining for the markers of mesothelial cells [14]. All experiments were performed using cells of the second to fifth cell passages. Tissue collection was approved by the Institutional Review Boards and informed consent was obtained from each patient.
Total RNA was extracted from HPMCs that were prepared by using TRIzol reagent (Gibco Invitrogen, Carlsbad, CA, USA). The RNA pellet was suspended in DNase/RNase-Free distilled water and stored at ŌłÆ70┬░C until subsequent analysis. Mineralocorticoid receptor oligonucleotide primers used for the polymerase chain reaction (PCR) were as follows: forward primer: 5ŌĆ▓-ACCAAGCATTCATGTTCAGGCACC-3ŌĆ▓ and reverse primer: 5ŌĆ▓-AGCTCCCGTCATCTGGTTCTTGTT-3ŌĆ▓. PCR was performed in 10mM TrisŌĆōHCl (pH 9.0), 40mM KCl, 1.5mM MgCl2, and 0.1┬ĀU Taq polymerase (Promega), in a final volume of 20┬Ā┬ĄL. Reactions were carried out in a DNA thermal cycler (PerkinŌĆōElmer, Boston, MA, USA). Following initial denaturation at 94┬░C for 5 minutes, mixtures were subjected 35 cycles at 94┬░C for 30 seconds, annealing at 55┬░C for 30 seconds, primer extension at 72┬░C for 45 seconds, and a final extension at 72┬░C for 10 minutes. PCR products were fractionated on 1% agarose gel, followed by staining with 0.5┬Ā╬╝g/mL ethidium bromide (Gibco BRL, Grand Island, NY, USA). The amount of PCR products was normalized with a housekeeping gene, GAPDH (forward primer: 5ŌĆ▓-ACCACAGTCCATGCCATCAC-3ŌĆ▓ and reverse primer: 5ŌĆ▓-TCCACCACCCTGTTGCTGTA-3ŌĆ▓).
To measure cell proliferation, HPMCs (104 cells/well) were incubated with aldosterone (1ŌĆō100nM) in 96-well plates. After incubation for 2ŌĆō7 days at 37┬░C, MTS reagent [3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxy methoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium, inner salt; Promega] was added, and the amount of metabolized formazine was measured at 490┬Ānm with an automated plate enzyme-linked immunosorbent assay reader (Molecular Devices, Sunnyvale, CA, USA) according to the manufacturerŌĆÖs instructions.
The morphology of HPMC cells was observed by an inverted phase contrast microscope (Axiovert 200; Carl Zeiss, Oberkochen, Germany) and the images were obtained using a digital camera (AxioCam HRC; Carl Zeiss). For staining, cells were washed with phosphate-buffered saline (PBS) and fixed in 4% paraformaldehyde in PBS (25 minutes at 20┬░C) and permeabilized with 1% Triton X-100 in PBS (15 minutes at 4┬░C). After washing with PBS, the cells were treated with 5% bovine serum albumin in PBS for 1 hour before incubation with primary antibodies specific for cytokeratin (1:50; DAKO Cytomation, Carpinteria, CA, USA) or ╬▒-smooth muscle actin (SMA, 1:100; Abcam, Cambridge, UK) in 5% bovine serum albumin overnight at 4┬░C. After washing with 0.2% Tween 20 in PBS, the cells were incubated with goat anti-mouse IgGŌĆōfluorescein-isothiocyanate-conjugated secondary antibody (Santa Cruz Biotechnology, Santa Cruz, CA, USA) for 1 hour at room temperature in the dark. The cell nuclei were counterstained with 4ŌĆ▓,6-diamidino-2-phenylindole. Immunofluorescence was visualized under an Axiovert 200 fluorescence microscope (Carl Zeiss) with 10├Ś0.3 numerical aperture and 20├Ś0.4 numerical aperture objectives equipped with an AxioCam HRC digital camera (Carl Zeiss). Digital photographs were obtained with Axiovision release 4.3 (Carl Zeiss) and merged images were produced using Photoshop version 10 (Adobe Systems, Toronto, ON, Canada).
Protein samples were isolated from cell lysate (30┬Ā╬╝g), and boiled (95┬░C, 5 minutes) after mixing in reducing buffer. Proteins were resolved on 10% sodium dodecyl sulfate polyacrylamide gels, and transferred to polyvinylidene difluoride membranes. Membranes were blocked by incubation in 5% w/v nonfat milk powder in Tris-buffered saline for 30 minutes at room temperature, followed by incubation with primary antibodies directed against the following antigens overnight at 4┬░C: E-cadherin (BD Pharmingen, San Diego, CA, USA), ╬▒-SMA (Abcam), S1004A (DAKO), extracellular-signal-regulated kinase (ERK1/2, Santa Cruz Biotechnology), phospho-ERK (Santa Cruz Biotechnology), p38 mitogen-activated protein kinase (MAPK) (p38, Santa Cruz Biotechnology) and phospho-p38 MAPK (Santa Cruz Biotechnology). After washing the blot with PBSŌĆōTween 20, blots were incubated with horseradish-peroxidase-conjugated secondary antibodies corresponding to each primary antibody, followed by enhanced chemiluminescence detection (Santa Cruz Biotechnology). Positive immunoreactive bands were quantified by densitometry and compared with the expression of human ╬▓-actin (Santa Cruz Biotechnology).
HPMCs were incubated with 10╬╝M 2ŌĆ▓,7ŌĆ▓-dichlorofluororescein diacetate (DCF-DA) (Invitrogen, Carlsbad, CA, USA) prior to exposure to aldosterone (10nM) and/or spironolactone (1╬╝M). Serial fluorescence was measured using a fluorescent plate reader at excitation 485┬Ānm and emission 535┬Ānm (Molecular Devices).
To investigate the signal transduction pathway responsible for aldosterone-induced EMT, HPMCs were pretreated with inhibitors of ERK (PD98059, 10╬╝M) or p38 MAPK (SB203580, 10╬╝M), and stimulated with aldosterone for 48 hours. We also examined the role of reactive oxygen species (ROS) in phenotypic transition of HPMCs by examining the effect of various antioxidants, N-acetyl cysteine (NAC, 5mM, ROS scavenger), apocynin [10╬╝M, an inhibitor of nicotinamide adenine dinucleotide phosphate (NADPH) oxidase], and rotenone (1╬╝M, an inhibitor of mitochondrial electron transfer chain subunit I) on aldosterone-induced EMT and activation of MAPK.
The ABI PRISM 7000 sequence detection system was used for determining the level of transcripts with SYBR Green I as a double-stranded DNA-specific dye (Applied Biosystems, Foster City, CA, USA). The PCR was performed in 5┬ĄM cDNA, 10┬ĄM SYBR Green PCR Master Mix, and 5pM sense and antisense primers of E-cadherin: (forward primer: 5ŌĆ▓-ACCCCTGTTGGTGTCTTT-3ŌĆ▓, reverse primer: 5ŌĆ▓-TTCGGGCTTGTTGTCATTCT-3ŌĆ▓), ╬▒-SMA: (forward primer: 5ŌĆ▓-GGGAATGGGACAAAAAGACA-3ŌĆ▓, reverse primer: 5ŌĆ▓-CTTCAGGGGCAACACGAA-3ŌĆ▓) for a final volume of 20┬ĄM per reaction. Optimal primer concentrations were determined by preliminary experiments. The comparative cycle threshold method was used to assess the relative mRNA expression levels of the target genes. For statistical analysis, all PCR procedures were replicated at least three times. The amount of PCR products was normalized with the housekeeping gene, ╬▓-actin, to determine the relative expression ratios for each mRNA in relation to the control group.
Data are shown as mean┬▒standard deviation. All statistical analyses were carried out using SPSS for Windows version 11.0 (SPSS, Inc., Chicago, IL, USA). Results were analyzed using a KruskalŌĆōWallis nonparametric test, StudentŌĆÖs t test or one-way analysis of variance for multiple comparisons. Statistical significance was determined when P was <0.05.
There was a constitutive expression of mineralocorticoid receptor (MR) in HPMCs. Fig. 1 shows the mRNA expression of MR in HPMCs isolated from three different patients.
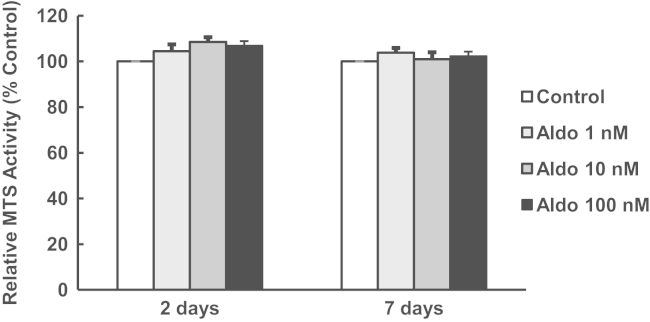
Stimulation of HPMCs with aldosterone (1ŌĆō100nM) for 2ŌĆō7 days did not induce an alteration of cell proliferation assessed by MTS assay (Fig. 2).
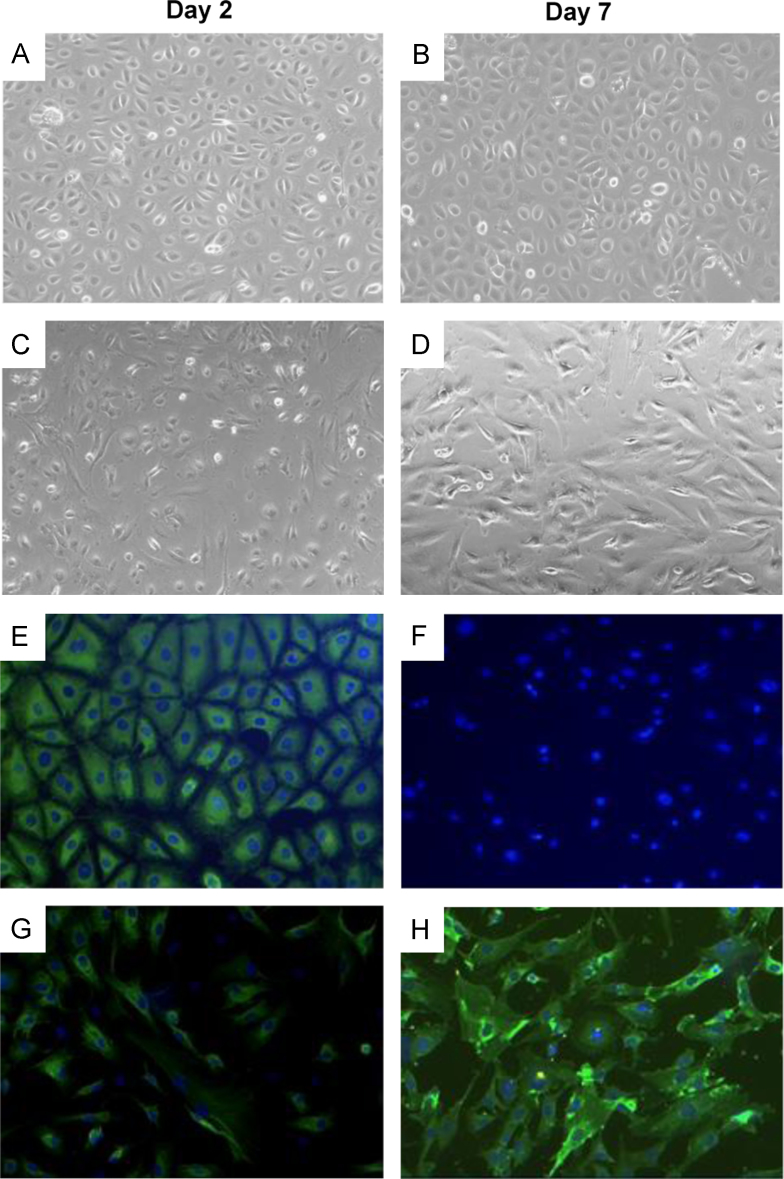
Aldosterone induced morphological changes in cultured HPMCs from Day 2 (Fig. 3). The typical cobblestone-shape of the HPMC monolayer disappeared on Day 2 of aldosterone stimulation with a change into an elongated morphology. After 7 days of aldosterone exposure, there was a loss of cell contact with a fibroblast-like phenotype. Immunofluorescent staining demonstrated a gradual decrease and redistrubution in cytokeratin and epithelial cell marker, and acquisition of ╬▒-SMA as early as 2 days after aldosterone stimulation (Fig. 3).
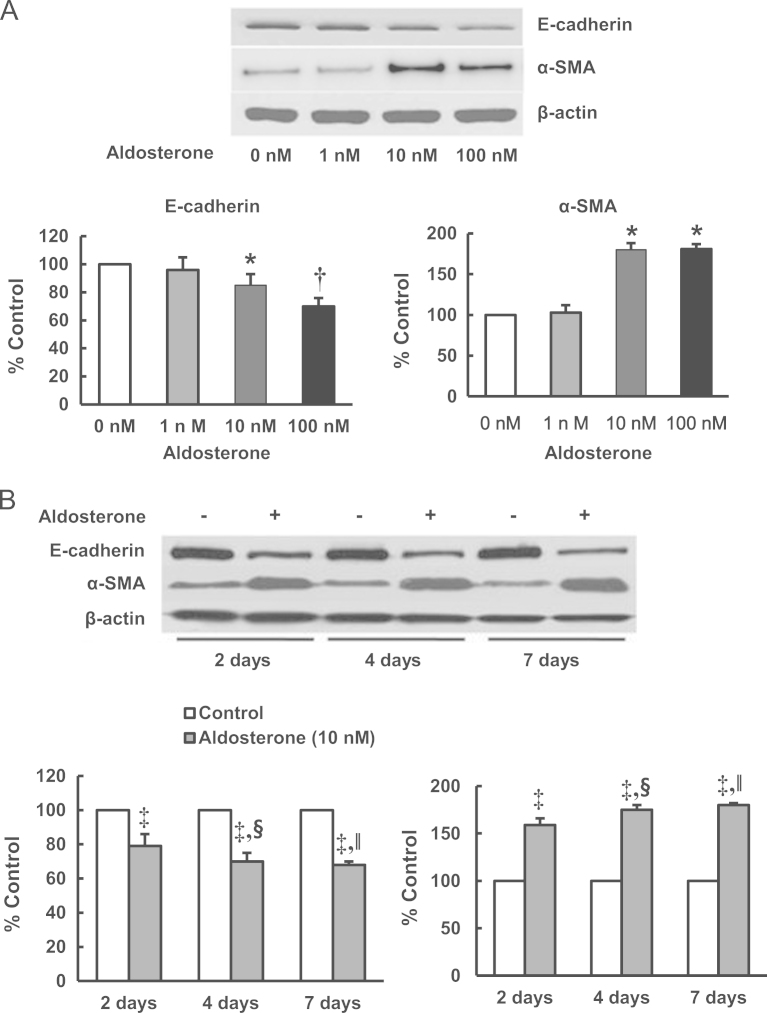
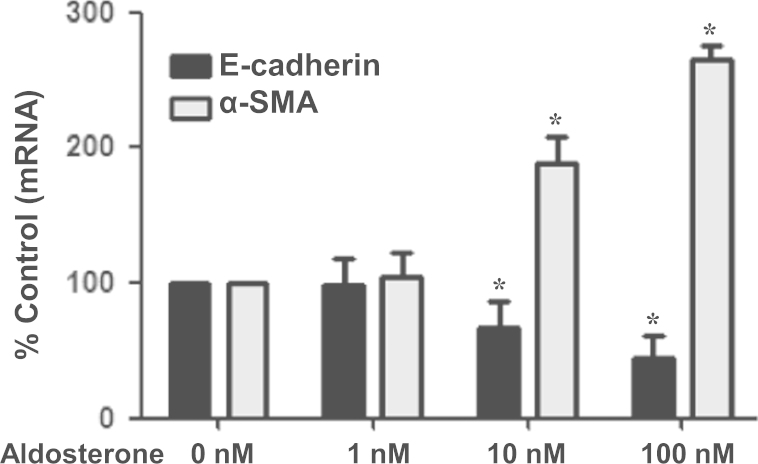
Exposure of HPMCs to aldosterone (1, 10 and 100nM) for 2ŌĆō7 days resulted in a dose- and time-dependent decrease in mRNA and protein expression of epithelial cell marker and E-cadherin, associated with an increase in expression of mesenchymal marker ╬▒-SMA (Figs. 4 and 5). The expression of S1004A, another specific marker of myofibroblasts, was increased by stimulation with aldosterone (Fig. 6). Aldosterone-induced changes in the expression of E-cadherin and ╬▒-SMA were almost completely inhibited by treatment with an MR antagonist, spironolactone (1╬╝M) (Fig. 7).
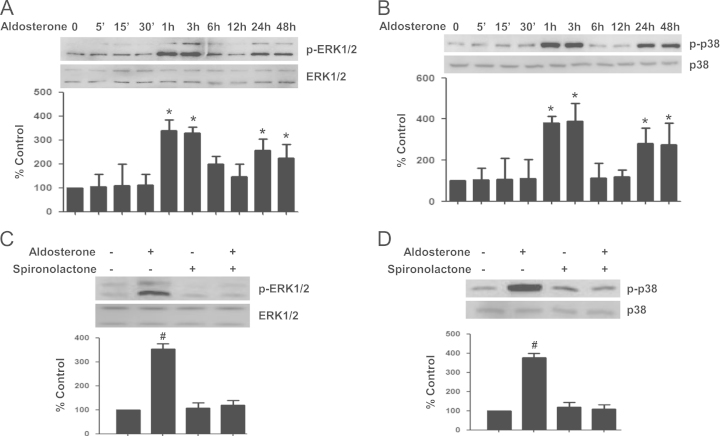
Aldosterone at a concentration of 10nM significantly increased phosphorylation of ERK1/2 from 1 hour of stimulation, which was blocked by spironolactone (Fig. 8). p38 MAPK was also activated by aldosterone at 1 hour, and showed a secondary peak of phosphorylation at 24 hours and 48 hours. Aldosterone-induced phosphorylation of p38 MAPK was inhibited by treatment with spironolactone.
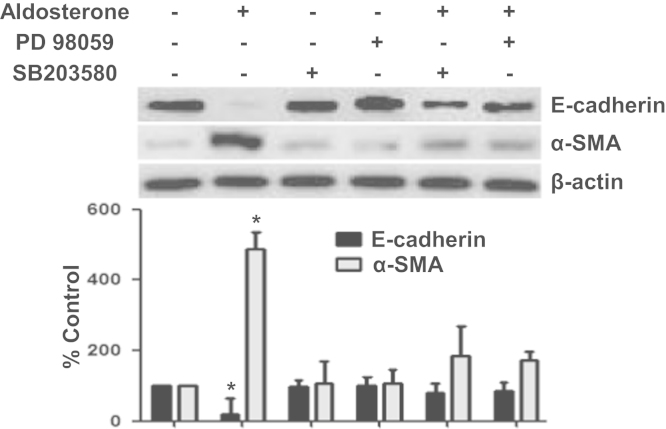
Both PD98059, an inhibitor of ERK pathway, and SB20358, an inhibitor of p38 MAPK, alleviated aldosterone-induced EMT of HPMCs (Fig. 9).
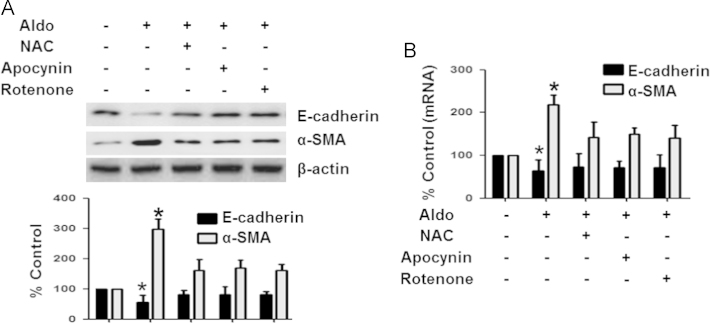
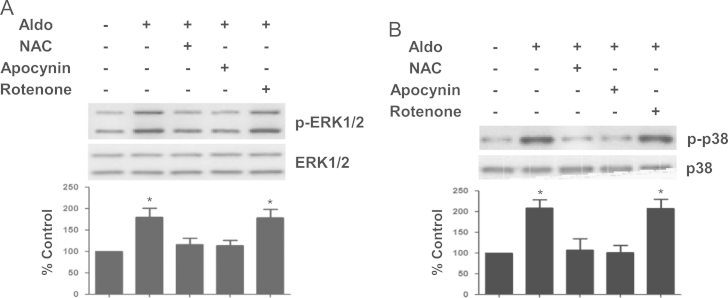
Aldosterone significantly increased the generation of intracellular ROS in HPMCs from 5 minutes (Fig. 10), which was blocked by spironolactone. NAC, apocynin and rotenone ameliorated E-cadherin downregulation and upregulation of ╬▒-SMA induced by aldosterone both at the protein and mRNA levels (Fig. 11). Aldosterone-induced ERK1/2 and p38 MAPK activation was ameliorated by NAC and apocynin, but not by rotenone (Fig. 12).
This study shows that aldosterone per se induces the phenotypic transition of HPMCs. The effect of aldosterone is MR dependent and involves intracellular ROS generation and activation of ERK1/2 and p38 MAPK. Antioxidants or MAPK inhibitors ameliorate aldosterone-induced EMT, suggesting these two mechanisms are responsible for aldosterone-induced changes in cell phenotype.
Long-term PD is frequently associated with functional and structural alterations in the peritoneal membrane [15,16]. Complex interactions of host and local factors, hypertonic glucose-based dialysate, acidity of lactate-buffered solution, and the presence of glucose degradation products with activation of inflammatory cytokines and various growth factors are known to be responsible for the changes in the peritoneal membrane [17,18]. Recent data suggest that EMT is an early and reversible step of peritoneal fibrosis [19]. EMT is a process by which epithelial cells lose their polarity and intercellular adhesion, and undergo remodeling of the intracellular cytoskeleton [20]. Concurrent with a loss of epithelial phenotype, cells undergoing EMT acquire the expression of mesenchymal components and manifest migratory and invasive properties [17,21,22]. Although EMT is a physiologically important process in embryogenesis and wound healing, it can impose unfavorable effect by promoting tissue fibrosis in nonphysiological conditions.
HPMCs possess their own reninŌĆōangiotensin system (RAS) with expression of angiotensinogen, angiotensin-converting enzyme, or angiotensin II type I receptor [23,24]. High glucose concentration results in RAS activation in HPMCs, and RAS blockers ameliorate the production of transforming growth factor-╬▓ and fibronectin, preserve ultrafiltration, and decrease peritoneal fibrosis [23]. As a continuum of previous studies, we found that HPMCs also constitutively express MR, indicating the presence of almost all components of the RAAS in peritoneal mesothelial cells. Profibrotic and proinflammatory effects of angiotensin II have been shown, along with demonstration of beneficial effects of angiotensin-converting enzyme inhibitor and angiotensin II type I receptor blocker on tubulointerstitial fibrosis or peritoneal fibrosis [25ŌĆō27]. Another end-product of the RAAS is aldosterone, a mineralocorticoid hormone, which is produced in the adrenal cortex, endothelial and vascular smooth muscle cells in the heart, blood vessels, and brain. Besides the classical effect on sodium and potassium transport, aldosterone is known to play a role in the development of cardiac and renal fibrosis. Chronic aldosterone infusion causes myocardial fibrosis in rats with high salt intake [28]. Similarly, aldosterone causes glomerular injury and tubulointerstitial fibrosis [29]. Previous studies also demonstrated that spironolactone decreases peritoneal thickening and inflammation in animal models of bacterial peritonitis and peritoneal fibrosis induced by mechanical scraping [30,31]. One study has investigated the effect of spironolactone (25┬Āmg) in a small number of PD patients (8 in placebo group vs. 8 in spironolactone group). Spironolactone administration for 6 months resulted in less inflammation and collagen deposition in peritoneal biopsy specimens [32]. However, these studies in animal models and PD patients neither address the mechanism of protective effect of spironolactone on peritoneal fibrosis nor focus on EMT as a potential mechanism of aldosterone-induced EMT. Our data suggest that blocking ROS generation with amelioration of ERK1/2 and p38 MAPK activation is the mechanism for the beneficial effect of spironolactone on aldosterone-induced EMT and fibrosis.
Aldosterone activated both ERK1/2 and p38 MAPK in HPMCs. Interestingly, aldosterone induced an activation of both MAPKs at 1ŌĆō3 hours, followed by secondary phosphorylation at 24ŌĆō48 hours. The significance of each peak of ERK and p38 activation needs to be further investigated. The initial activation of MAPK seems not to be related to the genomic effect of aldosterone, whereas the secondary peak may be the consequence of activation of genomic pathways by aldosterone. ERK1/2 and p38 MAPK are known to be important mediators of the intracellular signal transduction pathway responsible for collagen synthesis and tissue fibrosis [33]. ERK is implicated in cell proliferation and differentiation, whereas p38 mediates cellular stresses such as inflammation and death. A previous study demonstrated that angiotensin II-mediated EMT of renal tubular cells was ameliorated by MAPK inhibitors [34]. In our study, both ERK1/2 and p38 MAPK inhibitors alleviated aldosterone-induced EMT of HPMCs, indicating the role of MAPK in the development of EMT and peritoneal fibrosis.
In this study, intracellular ROS generation was one of the earliest findings of aldosterone-induced changes in HPMCs, which was observed after 5 minutes of aldosterone stimulation. There have been several studies demonstrating the role of ROS in the development of peritoneal fibrosis [35,36]. High glucose and glucose-based PD solutions induced generation of ROS in cultured HPMCs [37ŌĆō39]. Furthermore, treatment with antioxidants, NAC or catalase, effectively reversed high-glucose-induced change in E-cadherin and ╬▒-SMA [35]. The role of ROS in aldosterone-induced EMT was also examined in renal tubular cells [40]. In human proximal tubular cells, aldosterone increases intracellular ROS production and induced EMT. Aldosterone-induced EMT in renal tubular cells is completely blocked by mitochondrial respiratory chain complex I inhibitor [40]. In this study, we also found antioxidants including ROS scavenger, NADPH oxidase inhibitor, and mitochondria-targeted antioxidant ameliorated aldosterone-induced EMT of HPMCs, which was consistent with the previous study performed in renal tubular cells.
Importantly, ERK1/2 and p38 MAPK activation in aldosterone-stimulated HPMC was blocked by NAC and apocynin, but not by rotenone. This finding suggests ROS generation by NADPH oxidase precedes MAPK activation, which is followed by mitochondrial ROS generation. Mitochondrial ROS generation in HPMCs was observed after 6 hours of aldosterone stimulation (data not shown), in contrast to positive DCF-DA staining as early as after 5 minutes, which may have been due to activation of NADPH oxidase.
Aldosterone concentration used in this study was 0.1ŌĆō100nM. The normal plasma level of aldosterone is 0.14ŌĆō0.80nM in sitting and standing positions, and 0.08ŌĆō0.30nM in the lying position, which is lower than the levels that induced EMT in this study. However, in patients with kidney disease, liver cirrhosis, heart failure and a condition of increased intra-abdominal pressure, aldosterone level is increased [41]. Plasma level of aldosterone is reported to be higher in continuous ambulatory PD (CAPD) patients compared with other patients with chronic kidney disease. According to the previous study, mean plasma and dialysate level of aldosterone in 27 stable CAPD patients were 1.5┬▒0.6nM and 0.7┬▒0.3nM, respectively [42]. Higher plasma level of aldosterone in CAPD patients was considered to be due either to increased secretion or decreased clearance. The instillation of 2┬ĀL dialysate into the peritoneal cavity during CAPD may reduce hepatic blood flow and reduce the endogenous metabolic clearance rates of aldosterone, which results in a high level of aldosterone in peritoneal dialysate [43,44]. Although aldosterone concentrations inducing EMT in our in vitro study are higher than the aldosterone level in peritoneal dialysate, continuous exposure of peritoneal mesothelium to certain concentrations of aldosterone in PD patients may trigger phenotypic transition of mesothelial cells.
In conclusion, our study demonstrated that aldosterone induced EMT in HPMCs for the first time. Aldosterone-induced ROS generation plays a key role in aldosterone-induced EMT via activation of intracellular ERK1/2 and p38 MAPK. Our data suggest a potential therapeutic use of MR antagonist as well as MAPK inhibitor or specific antioxidants in peritoneal fibrosis.
Acknowledgments
This work was supported by the Bio & Medical Technology Development Program of the National Research Foundation (NRF) funded by the Ministry of Science, ICT & Future Planning (2010-0019866), the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIP) (2012R1A2A2A01013541) and the Korean Society of Nephrology (2008).
References
1. Krediet RT, Lindholm B, Rippe B. Pathophysiology of peritoneal membrane failure. Perit Dial Int 20(Suppl 4):2000;S22ŌĆōS42.


2. Miyata T, Devuyst O, Kurokawa K, van Ypersele de Strihou C. Toward better dialysis compatibility: advances in the biochemistry and pathophysiology of the peritoneal membranes. Kidney Int 61:2002;375ŌĆō386.


3. Williams JD, Topley N, Craig KJ, Mackenzie RK, Pischetsrieder M, Lage C, Passlick-Deetjen J; Euro Balance Trial Group. The Euro-Balance Trial: the effect of a new biocompatible peritoneal dialysis fluid (balance) on the peritoneal membrane. Kidney Int 66:2004;408ŌĆō418.


4. Yanez-Mo M, Lara-Pezzi E, Selgas R, Ram├Łrez-Huesca M, Dom├Łnguez-Jim├®nez C, Jim├®nez-Heffernan JA, Aguilera A, S├Īnchez-Tomero JA, Bajo MA, Alvarez V, Castro MA, del Peso G, Cirujeda A, Gamallo C, S├Īnchez-Madrid F, L├│pez-Cabrera M. Peritoneal dialysis and epithelial-to-mesenchymal transition of mesothelial cells. N Engl J Med 348:2003;403ŌĆō413.


6. R├╝ster C, Wolf G. ReninŌĆōangiotensinŌĆōaldosterone system and progression of renal disease. J Am Soc Nephrol 17:2006;2985ŌĆō2991.


7. Remuzzi G, Perico N, Macia M, Ruggenenti P. The role of renin-angiotensin-aldosterone system in the progression of chronic kidney disease. Kidney Int 99:2005;S57ŌĆōS65.

8. Brilla CG. ReninŌĆōangiotensinŌĆōaldosterone system and myocardial fibrosis. Cardiovasc Res 47:2000;1ŌĆō3.


9. Abbas G, Silveira MG, Lindor KD. Hepatic fibrosis and the renin-angiotensin system. Am J Ther 18:2011;e202ŌĆōe208.


10. Uhal BD, Li X, Piasecki CC, Molina-Molina M. Angiotensin signalling in pulmonary fibrosis. Int J Biochem Cell Biol 44:2012;465ŌĆō468.


11. Young MJ. Mechanisms of mineralocorticoid receptor-mediated cardiac fibrosis and vascular inflammation. Curr Opin Nephrol Hypertens 17:2008;174ŌĆō180.


12. Nishimura H, Ito Y, Mizuno M, Tanaka A, Morita Y, Maruyama S, Yuzawa Y, Matsuo S. Mineralocorticoid receptor blockade ameliorates peritoneal fibrosis in new rat peritonitis model. Am J Physiol Renal Physiol 294:2008;F1084ŌĆōF1093.


13. Stylianou E, Jenner LA, Davies M, Coles GA, Williams JD. Isolation, culture and characterization of human peritoneal mesothelial cells. Kidney Int 37:1990;1563ŌĆō1570.


14. Kang DH, Hong YS, Lim HJ, Choi JH, Han DS, Yoon KI. High glucose solution and spent dialysate stimulate the synthesis of transforming growth factor-beta1 of human peritoneal mesothelial cells: effect of cytokine costimulation. Perit Dial Int 19:1999;221ŌĆō230.


15. Williams JD, Craig KJ, Topley N, Von Ruhland C, Fallon M, Newman GR, Mackenzie RK, Williams GT; Peritoneal Biopsy Study Group. Morphologic changes in the peritoneal membrane of patients with renal disease. J Am Soc Nephrol 13:2002;470ŌĆō479.


16. Plum J, Hermann S, Fussholler A, Schoenicke G, Donner A, R├Čhrborn A, Grabensee B. Peritoneal sclerosis in peritoneal dialysis patients related to dialysis settings and peritoneal transport properties. Kidney Int Suppl 78:2001;S42ŌĆōS47.


17. Margetts PJ, Bonniaud P. Basic mechanisms and clinical implications of peritoneal fibrosis. Perit Dial Int 23:2003;530ŌĆō541.


18. Kim YL. The challenge of PD patients: glucose and glucose degradation products in PD solution. Kidney Res Clin Pract 31:2012;A46ŌĆōA47.

19. Jang YH, Shin HS, Sun Choi H, Ryu ES, Jin Kim M, Ki Min S, Lee JH, Kook Lee H, Kim KH, Kang DH. Effects of dexamethasone on the TGF-beta1-induced epithelial-to-mesenchymal transition in human peritoneal mesothelial cells. Lab invest 93:2013;194ŌĆō206.


20. Aguilera A, Yanez-Mo M, Selgas R, S├Īnchez-Madrid F, L├│pez-Cabrera M. Epithelial to mesenchymal transition as a triggering factor of peritoneal membrane fibrosis and angiogenesis in peritoneal dialysis patients. Curr Opin Investig Drugs 6:2005;262ŌĆō268.

21. Selgas R, Bajo A, Jimenez-Heffernan JA, Del Peso G, Aguilera A, L├│pez-Cabrera M. Epithelial-to-mesenchymal transition of the mesothelial cell ŌĆō its role in the response of the peritoneum to dialysis. Nephrol Dial Transplant 21(Suppl 2):2006;ii2ŌĆōii7.


22. Aroeira LS, Aguilera A, S├Īnchez-Tomero JA, Bajo MA, del Peso G, Jim├®nez-Heffernan JA, Selgas R, L├│pez-Cabrera M. Epithelial to mesenchymal transition and peritoneal membrane failure in peritoneal dialysis patients: pathologic significance and potential therapeutic interventions. J Am Soc Nephrol 18:2007;2004ŌĆō2013.


23. Noh H, Ha H, Yu MR, Kim YO, Kim JH, Lee HB. Angiotensin II mediates high glucose-induced TGF-beta1 and fibronectin upregulation in HPMC through reactive oxygen species. Perit Dial Int 25:2005;38ŌĆō47.


24. Sauter M, Cohen CD, Wornle M, Mussack T, Ladurner R, Sitter T. ACE inhibitor and AT1-receptor blocker attenuate the production of VEGF in mesothelial cells. Perit Dial Int 27:2007;167ŌĆō172.


25. Basturk T, Unsal A, Koc Y, Nezaket E, Ahbap E, Sakaci T, Sevinc M. The effects of angiotensin-converting enzyme inhibitors on peritoneal protein loss and solute transport in peritoneal dialysis patients. Clinics (Sao Paulo) 67:2012;877ŌĆō883.



26. Sun J, Yu K, Zhang H, Qun W, Rong W. Effect of renin-angiotensin system inhibitors on prevention of peritoneal fibrosis in peritoneal dialysis patients. Nephrology 15:2010;27ŌĆō32.

27. Whaley-Connell A, Habibi J, Panfili Z, Hayden MR, Bagree S, Nistala R, Hyder S, Krueger B, Demarco V, Pulakat L, Ferrario CM, Parrish A, Sowers JR. Angiotensin II activation of mTOR results in tubulointerstitial fibrosis through loss of N-cadherin. Am J Nephrol 34:2011;115ŌĆō125.



28. Brilla CG, Weber KT. Mineralocorticoid excess, dietary sodium, and myocardial fibrosis. J Lab Clin Med 120:1992;893ŌĆō901.

29. Greene EL, Kren S, Hostetter TH. Role of aldosterone in the remnant kidney model in the rat. J Clin Invest 98:1996;1063ŌĆō1068.



30. Ersoy R, Celik A, Yilmaz O, Sarioglu S, Sis B, Akan P, Yenicerioglu Y, Ormen M, Camsari T. The effects of irbesartan and spironolactone in prevention of peritoneal fibrosis in rats. Perit Dial Int 27:2007;424ŌĆō431.


31. Zhang L, Hao JB, Ren LS, Ding JL, Hao LR. The aldosterone receptor antagonist spironolactone prevents peritoneal inflammation and fibrosis. Lab Invest 94:2014;839ŌĆō850.


32. Vazquez-Rangel A, Soto V, Escalona M, Toledo RG, Castillo EA, Polanco Flores NA, Falcon-Chavez I, Madero M. Spironolactone to prevent peritoneal fibrosis in peritoneal dialysis patients: a randomized controlled trial. Am J Kidney Dis 63:2014;1072ŌĆō1074.


33. Bakin AV, Rinehart C, Tomlinson AK, Arteaga CL. p38 mitogen-activated protein kinase is required for TGFbeta-mediated fibroblastic transdifferentiation and cell migration. J Cell Sci 115:2002;3193ŌĆō3206.


34. Chen J, Chen JK, Harris RC. Angiotensin II induces epithelial-to-mesenchymal transition in renal epithelial cells through reactive oxygen species/Src/caveolin-mediated activation of an epidermal growth factor receptor-extracellular signal-regulated kinase signaling pathway. Mol Cell Biol 32:2012;981ŌĆō991.



35. Noh H, Kim JS, Han KH, Lee GT, Song JS, Chung SH, Jeon JS, Ha H, Lee HB. Oxidative stress during peritoneal dialysis: implications in functional and structural changes in the membrane. Kidney Int 69:2006;2022ŌĆō2028.


36. Fukuoka N, Sugiyama H, Inoue T, Kikumoto Y, Takiue K, Morinaga H, Nakao K, Maeshima Y, Asanuma M, Wang DH, Ogino K, Masuoka N, Makino H. Increased susceptibility to oxidant-mediated tissue injury and peritoneal fibrosis in acatalasemic mice. J Nephrol 28:2008;661ŌĆō668.

37. Lee H, Yu MR, Song JS, Ha H. Reactive oxygen species amplify protein kinase C signaling in high glucose-induced fibronectin expression by human peritoneal mesothelial cells. Kidney Int 65:2004;1170ŌĆō1179.


38. Ha H, Lee H. Effect of high glucose on peritoneal mesothelial cell biology. Perit Dial Int 293(20):2000;23ŌĆō26.

39. Hung KY, Liu SY, Yang TC, Liao TL, Kao SH. High-dialysate-glucose-induced oxidative stress and mitochondrial-mediated apoptosis in human peritoneal mesothelial cells. Oxid Med Cell Longev 64:2014;2793ŌĆō2805.

40. Zhang A, Jia Z, Guo X, Yang T. Aldosterone induces epithelial-mesenchymal transition via ROS of mitochondrial origin. Am J Physiol Renal Physiol 293:2007;F723ŌĆōF731.


41. Bloomfield GL, Blocher CR, Fakhry IF, Sica DA, Sugerman HJ. Elevated intra-abdominal pressure increases plasma renin activity and aldosterone levels. J Trauma 42:1997;997ŌĆō1004.


42. Selgas R, Munoz IM, Conesa J, Madero R, Gancedo PG, Carmona AR, Eugenia M, Huarte ME, Fontan MP, Sicilia LS. Endogenous sympathetic activity in CAPD patients: its relationship to peritoneal diffusion capacity. Perit Dial Int 6:1986;205ŌĆō208.

Figure┬Ā1
Expression of mineralocorticoid receptor mRNA in HPMCs. Mineralocorticoid receptor mRNA detected in cultured HPMCs. Representative reverse transcriptase polymerase chain reaction bands from three different patients are shown. NC (negative control) denotes the sample containing all reactants except cDNA. HPMC, human peritoneal mesothelial cell; MR, mineralocorticoid receptor.

Figure┬Ā2
Effect of aldosterone on the proliferation of human peritoneal mesothelial cells. There was no effect of aldosterone (1ŌĆō100nM) on MTS activity on Day 2 and 7. Data are presented as means┬▒standard deviation. Aldo, aldosterone; MTS, 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxy methoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium, inner salt.
Figure┬Ā3
Effects of aldosterone on morphology and expression of cytokeratin and ╬▒-SMA in HPMCs. Compared with the cobblestone appearance of unstimulated HPMCs on Day 2 (A) and Day 7 (B), aldosterone (10nM) induced phenotypic transformation of cells. Cells began to elongate and acquire a fibroblast-like morphology after 2 days of aldosterone stimulation (C), which was further evident after 7 days (D). Immunofluorescence staining for cytokeratin revealed abundant cytoplasmic expression in unstimulated cells (E), which was markedly decreased upon aldosterone stimulation (G). Aldosterone upregulated ╬▒-smooth muscle actin organization (H) in contrast with almost negative staining in unstimulated cells (F). Magnification, 100├Ś. HPMC, human peritoneal mesothelial cell; SMA, smooth muscle actin.
Figure┬Ā4
Effects of aldosterone on the expression of E-cadherin and ╬▒-SMA in HPMCs. Aldosterone induced a dose-dependent decrease in expression of E-cadherin and increase in ╬▒-SMA expression after 2 days stimulation (A). Aldosterone (10nM) also induced time-dependent changes in expression of E-cadherin and ╬▒-SMA between 2 days and 7 days (B). Representative western blots with a quantitation bar graph are shown. ŌüÄP<0.05 versus control (0nM) or 1nM aldosterone, ŌĆĀP<0.05 versus 10nM aldosterone, ŌĆĪP<0.05 versus control at each time point, ┬¦P<0.05 versus aldosterone at 2 days, ||P<0.05 versus aldosterone at Day 2 and Day 4 (n=5). HPMC, human peritoneal mesothelial cell; SMA, smooth muscle actin.
Figure┬Ā5
Effects of aldosterone on mRNA expression of E-cadherin and ╬▒-SMA in HPMCs. Aldosterone induced a significant decrease in the mRNA expression of E-cadherin and an increase in ╬▒-SMA at concentrations >10nM after 2 days. ŌüÄP<0.05 versus 0 and 1nM aldosterone (n=4). HPMC, human peritoneal mesothelial cell; SMA, smooth muscle actin.
Figure┬Ā6
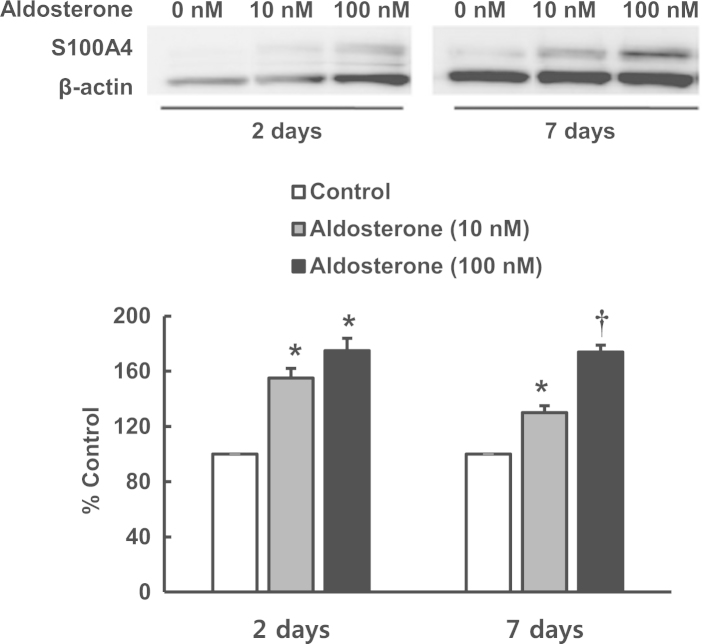
Effect of aldosterone on expression of S100A4 in HPMCs. Aldosterone (10ŌĆō100nM) increased expression of S100A4 protein on Day 2 and Day 7 in a dose-dependent manner. Representative western blots with a quantitation bar graph are shown. ŌüÄP<0.05 versus control (0nM) at each time point, ŌĆĀP<0.05 versus control and 10nM aldosterone (n=3). HPMC, human peritoneal mesothelial cell.
Figure┬Ā7
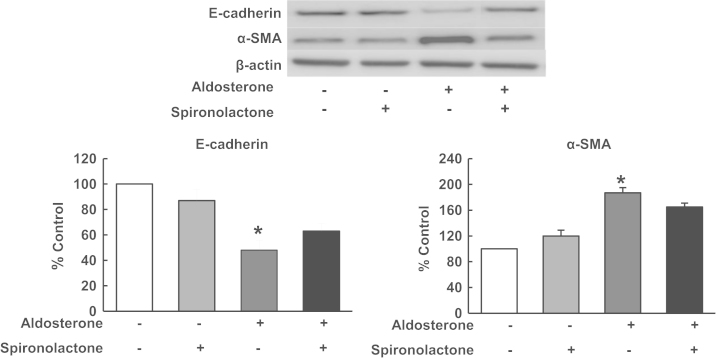
Effect of spironolactone on aldosterone-induced epithelial-to-mesenchymal transition in HPMCs. Aldosterone (10nM)-induced changes in expression of E-cadherin and ╬▒-SMA were blocked by pretreatment with spironolactone (1╬╝M). Representative western blots with a quantitation bar graph are shown. ŌüÄP<0.05 versus others (n=5). HPMC, human peritoneal mesothelial cell; SMA, smooth muscle actin.
Figure┬Ā8
Effects of aldosterone on the activation of ERK1/2 and p38 MAPK in HPMCs with or without spironolactone pretreatment. Aldosterone (10nM) significantly increased phosphorylation of ERK1/2 (A) and p38 MAPK (B) in HPMCs. Aldosterone-induced activation of ERK1/2 and p38 MAPK was inhibited by spironolactone (1╬╝M) (C, D). Representative western blotting and its quantitation graphs are shown (n=4). ŌüÄP<0.05 versus others. ERK, extracellular signal-regulated kinase; HMPC, human peritoneal mesothelial cell; MAPK, mitogen-activated protein kinase.
Figure┬Ā9
Effect of MAPK inhibitors on aldosterone-induced EMT. ERK 1/2 inhibitor (PD98059, 10╬╝M) and p38 inhibitor (SB203589, 10╬╝M) alleviated the decrease of E-cadherin and acquisition of ╬▒-SMA in aldosterone-exposed cells at 48 hours. Representative western blotting and its quantitation graphs are shown (n=6). ŌüÄP<0.05 versus others. EMT, epithelial-to-mesenchymal transition; ERK, extracellular signal-regulated kinase; MAPK, mitogen-activated protein kinase; SMA, smooth muscle actin.
Figure┬Ā10
Effect of spironolactone on aldosterone-induced ROS production. Aldosterone (10nM) significantly increased generation of intracellular ROS in human peritoneal mesothelial cells after 5 minutes; the effect was blocked by co-treatment with spironolactone (1╬╝M). Representative DCF-DA staining at 30 minutes of aldosterone treatment is shown (n=5). ŌüÄP<0.05 versus others. Aldo, aldosterone; DCF-DA, 2ŌĆ▓,7ŌĆ▓-dichlorofluororescein diacetate; ROS, reactive oxygen species.

Figure┬Ā11
Effects of antioxidants on aldosterone-induced EMT. Aldosterone-induced changes in expression of E-cadherin and ╬▒-SMA were blocked by NAC (5mM), apocynin (10╬╝M), and rotenone (5╬╝M) (n=5). Representative western blotting with its quantitation graph (A) and relative mRNA expression (B) are shown. ŌüÄP<0.05 versus others. Aldo, aldosterone; EMT, epithelial-to-mesenchymal transition; NAC, N-acetyl cysteine; SMA, smooth muscle actin.
Figure┬Ā12
Effect of antioxidants on aldosterone-induced MAPK activation. Aldosterone (10nM) significantly increased phosphorylation of ERK1/2 (A) and p38 MAPK (B) in human peritoneal mesothelial cells. Aldosterone-induced activation of ERK1/2 and p38 MAPK was significantly ameliorated by antioxidants, NAC (5mM) or apocynin (10╬╝M). However, rotenone (5╬╝M) did not alter the aldosterone-induced activation of ERK1/2 and p38 MAPK. Representative western blotting and its quantitation graphs are shown (n=4). ŌüÄP<0.05 versus others. Aldo, aldosterone; ERK, extracellular signal-regulated kinase; MAPK, mitogen-activated protein kinase; NAC, N-acetyl cysteine.



 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print
















